The importance of data in assessing the efficacy of biostimulants
Background: As the need for biostimulants becomes better understood and their place in the armoury of agronomists more established, creating a credible means of generating and assessing their use and efficacy becomes more important. Maxstim Ltd is a leader in biostimulant design and manufacture and understands the need to demonstrate the integrity and quality of the supporting data that underpins the performance of our products.
This document describes in detail how our product development and technical team carry out the assessment of product performance and how the efficacy data is generated and assessed for quality and reporting. At the same time recognising that having solid data on product effects is irrelevant if it makes no contribution to real farmer/grower value.
Maxstim procedures for supporting biostimulant claims
In order to enable access to the European Union single market, CE marking is required for all fertilising products (the definition of which now includes biostimulants).
This is covered by Regulation (EU) 2019/1009 FPR published in July 2019 and which will be fully operational in July 2022.
The European Biostimulant Industry Council (EBIC) have developed some guidance to follow when justifying plant biostimulant claims. These principles are expected to be incorporated into harmonized European standards currently being developed by the European Committee for Standardization (CEN) to support the implementation of the regulation (Ricci et al., 2019; General principles to justify plant biostimulant claims. Frontiers in Plant Science Volume 10, article 494).
Maxstim has adopted these procedures for evidencing biostimulant claims. These are based on the EU definition of biostimulants:
An EU fertilising product the function of which is to stimulate plant nutrition processes independently of the product’s nutrient content with the sole aim of improving one or more of the following characteristics of the plant or the plant rhizosphere:
(a) nutrient use efficiency
(b) tolerance to abiotic stress
(c) quality traits
(d) availability of confined nutrients in the soil or rhizosphere
(e) yield improvement (yield increase or yield security)
Note that the effects of biotic stress cannot be entirely separated from abiotic stress. Consequently, reducing abiotic stress can have an indirect effect on the impact of pests and diseases on crop plants.
Plant Biostimulants “shall have effects that are claimed on the label”
- A conformity assessment dossier will be compiled for Maxstim products and include evidence of the product’s effects
- Harmonized European Standards will indicate what constitutes compelling evidence
Underlying Principles to Justify a Biostimulant Claim: Data
- Existing data (pre Regulation 2019/1009 FPR) can be used
- Existing data sets and new data should comply with acceptable Experimental Quality Criteria (this can be generated by the manufacturer, independent researchers, distributors or growers)
- Data should have agronomic relevance (i.e. yield improvement, must demonstrate a biologically relevant agronomic trend)
Data should be of sufficient quality to support any claim:
To assess data quality a Klimisch score (Klimisch et al., 1997) can be used to evaluate data robustness. Scores of 1 and 2 are acceptable, though data with scores of 3 and 4 can be used as part of a wider assessment e.g. supporting observations of consistent “agronomically” positive data trends (i.e., not necessarily statistically significant) compared to untreated plots in field trials.
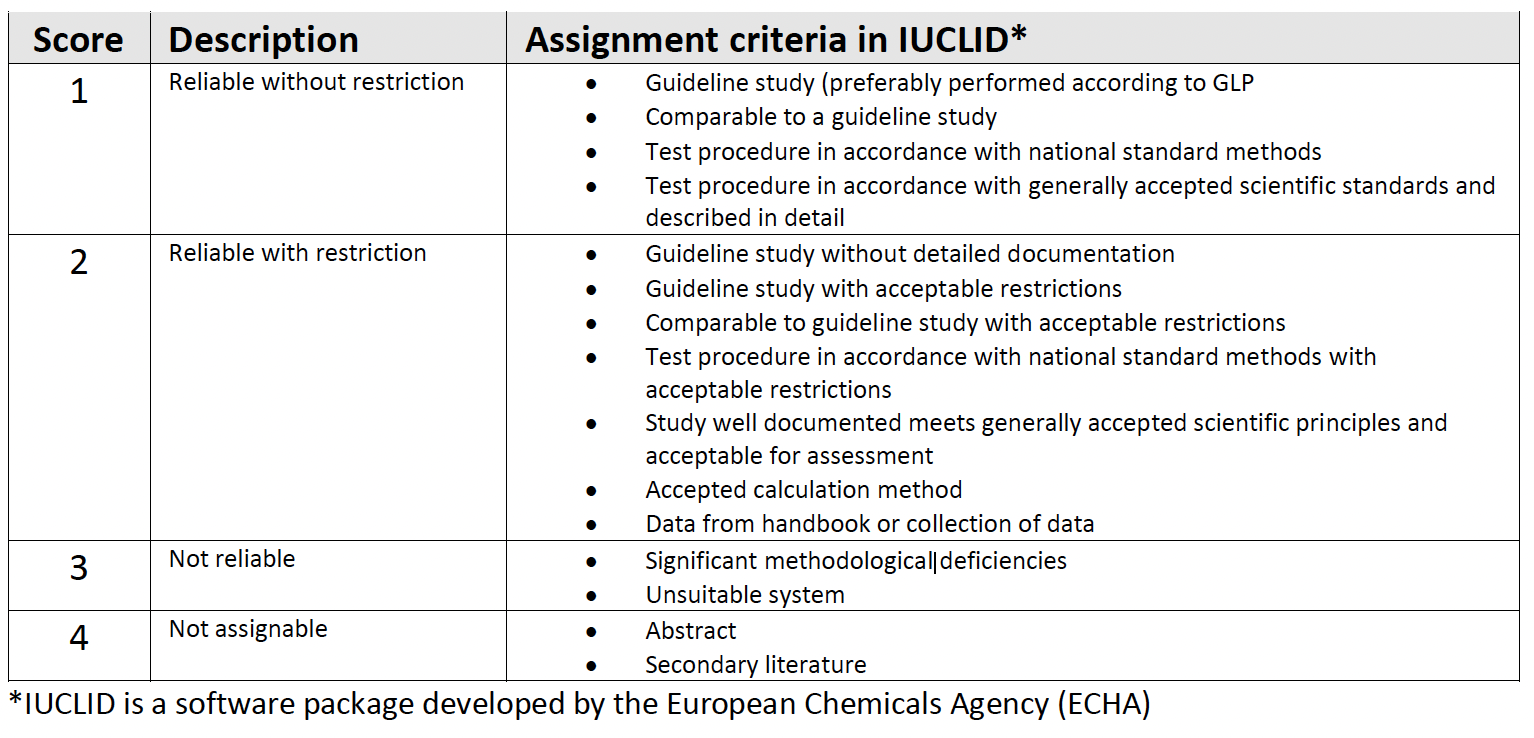
For new data:
Data should be of acceptable experimental quality and data should include:
- Qualifications of those undertaking the study
- A test/experimental plan
- Who did what, when and why?
Agronomic relevance
- Having solid data on product effects is irrelevant if it makes no contribution to real farmer/grower value
- However, value is sometimes only measurable under certain conditions (i.e. the effects of increased root growth on water uptake may only be visible where water is restricted)

